Fuel Cells (Hydrogen, Ethanol and Solid)
Call for specifications and pricing
Standard 2 Year Warranty
Extended warranties available
Fuel Cells Suppliers
Types of Fuel Cells.
Most fuel cells are categorised by the;
- Type of electrolyte they use
- Kind of catalysts required
- Temperature range in which they operate
- Type of fuel required
And of course other factors too.
These characteristics determine the type of application for which the cell can be used. Each type of fuel cell is developed to offer its own benefits and limitations. See more about the following fuel cell types below.
Please Note: While the information below is provided to be helpful and informative, iWolf’s fuel cell portfolio is ever changing in alignment with technology trends, availability and prices. Please call for the latest pricing, specifications and installation options on our complete range of fuel cells.
Polymer Electrolyte Membrane (PEM) fuel cells
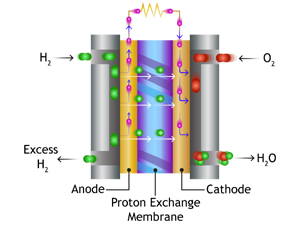
Polymer electrolyte membrane (PEM) fuel cells are also referred to as proton exchange membrane fuel cells. They deliver high power density, low weight and volume compared with many other fuel cells.
PEM fuel cells use a solid polymer as an electrolyte and porous carbon electrodes containing a platinum or platinum alloy catalyst. They require only hydrogen, oxygen and water to function.
PEM fuel cells run at comparatively low temperatures, around 80°C. This low-temperature operation allows them to start with less warm-up time and causes less wear on components making it far more durable.
On the down side, the PEM cell is dependent on a noble-metal catalyst which is usually made from platinum thus adding to the total system cost.
Platinum catalysts are extremely sensitive to carbon monoxide poisoning so an additional reactor is required to reduce carbon monoxide in the fuel gas if the hydrogen is made from a hydrocarbon fuel, also adding to the cost of manufacture.
Ideal for transportation applications such as cars and buses and some stationary applications PEM cells are a popular choice.
Direct Methanol (DMFCs) fuel cells
Direct methanol fuel cells are powered by pure methanol which is often mixed with H2O and injected directly into the fuel cell anode.
DMFCs do not have as many fuel storage problems that is typical in some fuel cell systems.
This is because methanol has a greater energy capacity than hydrogen. Methanol is easier to transport and supply using todays current infrastructure.
Direct methanol Fuel cell applications are typically used to provide power for portable fuel cell applications like mobile phones and laptop computers.
Alkaline fuel cells (AFCs)
One on the first fuel cell technologies ever developed, the Alkaline fuel cells (AFCs) was the first type widely used in America’s space program to produce electrical power and water on-board the craft.
AFCs use a solution of potassium hydroxide in H2O as the electrolyte and a variety of non-precious metals as the catalyst at the anode and cathode.
Some AFCs today have been developed to incorporate a polymer membrane as the electrolyte. Closely related to standard PEM fuel cell, these cells use an alkaline membrane instead of an acid membrane.
The AFCs high performance is due to the faster speed at which the electro-chemical reactions occur inside the cell. They have proven efficiencies well above 60% in the outer space.
Alkaline fuel cells incorporating liquid electrolytes can run in “recirculating mode” which promotes electrolyte regeneration reducing the impact of carbonate formation within the electrolyte which can introduces issues with shunt currents. Liquid electrolyte systems also suffer from “wettability” which increases corrosion and problems handling differential pressures.
Alkaline Membrane fuel cells (AMFCs)
The development of AMFCs have averted the concerns with AFCs and are less vulnerable to CO2 poisoning than liquid-electrolyte AFCs are.
However, CO2 still affects performance and durability of AMFCs and still lack that of PEMFCs. AMFCs are a good choice for applications requiring Watts to Kilowatts scalable capacity.
Phosphoric Acid fuel cells (PAFCs)
Phosphoric acid fuel cells incorporate liquid phosphoric acid as the electrolyte. The acid is controlled in a Teflon-bonded silicon carbide matrix with porous carbon electrodes containing a platinum catalyst.
PAFC, the “Next Gen” fuel cells is a mature cell type and the first to be used commercially. Typically used for stationary power generation, PAFCs have been used to power large vehicles such as trains and buses.
PAFC efficiency is only slightly higher than that of combustion-based power plants which operate at around 33%. PAFCs are also less powerful than other fuel cells, given the same weight and volume. As a result, these fuel cells are typically large and heavy.
Molten Carbonate fuel cells (MCFCs)
MCFCs are high-temperature fuel cells. They use an electrolyte comprised of a molten carbonate salt mixture in a porous, inert ceramic lithium aluminium oxide matrix. As they operate at high temperatures of 650°C, non-precious metals can be used as catalysts at the anode and cathode.
MCFCs improved efficiency offers a significant cost reduction over phosphoric acid fuel cells. Molten carbonate fuel cells can reach efficiencies approaching 65% when coupled with a turbine. Waste heat can also be captured and reused increasing the overall fuel efficiency to over 85%.
As MCFCs operate at high temperatures, methane and other light hydrocarbons are converted to hydrogen inside the fuel cell through a process called internal reforming which also increases efficiency.
Solid Oxide fuel cells (SOFCs)
Solid oxide fuel cells incorporate a hard, non-porous ceramic compound as the electrolyte. SOFCs are around 60% efficient when converting fuel to electricity. In waste heat capture co-generation applications the overall efficiencies can top 85%.
1) SOFCs are the most sulfur-resistant fuel cell type and are not poisoned by carbon monoxide which can also be used as fuel. This technology allows SOFCs to use natural gas, biogas and other gases made from coal.
2) SOFCs run at very high temperatures, around 1,000°C. As explained previously, high-temperature operation eliminates the need for a precious-metal catalyst and therefore reducing cost.
3) SOFCs can also reform fuels internally enabling them to use a variety of fuels while reducing the cost of adding a reformer to the system.
Reversible fuel cells (RFCs)
Like several other fuel cell types, reversible fuel cells can produce electricity from hydrogen and oxygen to generate heat and water as by-products.
Reversible fuel cell systems can also use electricity from, hydro power, wind power solar power or other forms to split water back into oxygen and hydrogen fuel through electrolysis.
Reversible fuel cells can also store the excess energy from these power sources in the form of hydrogen. This energy storage capability the key to intermittent renewable energy technologies.